I recently learned that there is a *maximum* age at which one can start training to be an air traffic controller. While a minimum age for various efforts is common, specifying a maximum age seemed curious, and especially given that the oldest you can be to start ATC training is 30 years old. So young!
Naturally, I wondered why this limit had been chosen. After some digging, I discovered that it derives from studies done in the 1960s and 1970s such as
Trites and Cobb (1962) conducted a study of ATC trainees and their subsequent job performance (in the first year of work) that showed a marked increase in training failure rates with age, up to age 45:
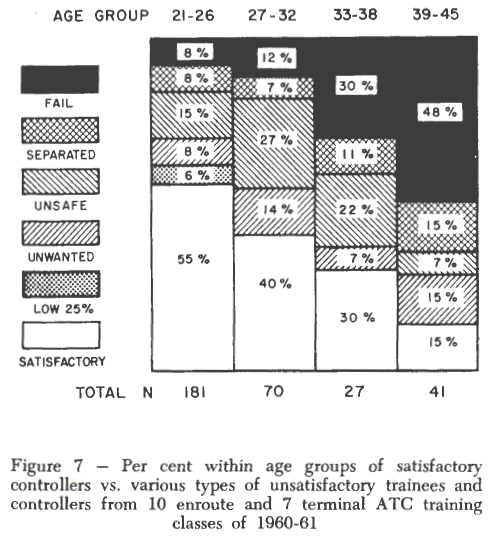
They do not speculate about reasons for the reduction in performance, concluding that
“Whatever the nature of the casual factors associated with chronological age and underlying the relationships of this study, there is no doubt that the number of potential training failures can be reduced and undesirable controllers eliminated by specifying a maximum age for entry into air traffic controller training. In the best interests of air safety and financial economy, establishment of an upper age limit is recommended.”
The FAA must not have heeded this advice, because nine years later, Cobb was still working to persuade them of the dangers of older ATC trainees. The Cobb et al. (1971) study is of 710 air traffic controllers, aged 21-52, that concluded that “age correlated negatively with 21 of the 22 aptitude measures and with training course grades.” This is a study of a biased sample, however: “because of their highly specialized pre-employment experience, these men were not required to qualify on the CSC ATC Aptitude Screening Test.” It is perhaps unsurprising that they might have lower aptitude measures, since these were not used to screen them as applicants. However, the negative correlation of performance with age is there. In Figure 2 from this paper, black means “failed basic training course”, hashed means “course grades comprising the approximate lower half of pass group”, and white means “course grades comprising the approximate upper half of pass group”:

The numbers in the right column are the number of subjects in each age group. “Although the subjects over age 34 represented only about 23 per cent of the 710 men involved in the entire study, their failure rate (31.1 per cent) in Academy ATC training was about three times that of the younger trainees.”
Cobb et al. went on to test these subjects on a variety of mental tasks, including simple arithmetic, spatial reasoning, following oral directions, abstract/logical reasoning, and a job-relevant task described as follows: “A highly-speeded test consisting of two parts of thirty items each. In each part, the subject is presented a flight data display for several aircraft and must determine whether certain changes in altitude may be directed without violating a specified time-separation rule.”
Performance on every single test, except arithmetic, was negatively correlated with age.
Maybe this result, or others like it, did the trick. The right of the FAA to establish a maximum age for its air traffic controllers was passed into US Law in 1972. The current version of the law states that
“The Secretary may, with the concurrence of such agent as the President may designate, determine and fix the maximum limit of age within which an original appointment to a position as an air traffic controller may be made.”





