February 7th, 2013 at 9:46 pm (Animals, Biology)
I killed two ants the other day, for Science. “Adventures with a Microscope” instructs you in how to kill a fly so that you can remove its legs and eyes for a close look under the microscope. No flies were available, so I victimized some ants who kept trying, with mindless persistence, to invade my personal space. I put them under glass, where one asphyxiated, slowly, as I had no chloroform. The other escaped and I flicked it into some water, where it drowned. I felt more than a few twinges of guilt, staring at their huddled corpses, but then decided to examine them under the microscope.
And oh, how glorious!
40x (lowest magnification):
The segmented, slightly blurry object on the right is its antenna.
The thin filament is (I think) carpet fuzz.
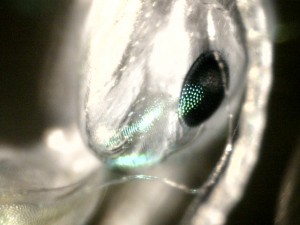
Note the difference in texture between chitin and eye (zoom 100x):

Zoom 200x:

These were all taken with reflected light, since ants don’t transmit light well. But are they not beautiful and alien, seen up so close? We’ve all heard of the fly’s multi-faceted eye. I’m not sure I realized that the ant’s is likewise complex and compound.
Wow.
7 Comments
4 of 5 people learned something from this entry.
January 22nd, 2013 at 11:33 pm (Biology, Chemistry)
Adventure 2 in “Adventures with a Microscope” is titled “We Become Crystal Gazers.” The author continues:
We are not going to peer into such crystals as fortune tellers use, in which they claim to be able to predict that some rich relative is going to leave you money, or some equally nonsensical bosh.
No indeed, this is about chemistry. So I followed along, heated some water, and mixed up supersaturated solutions of various interesting substances from my kitchen. And wow, check it out! (Click to enlarge.)

Salt. Best viewed ~1 hour after deposition on the slide, with partial crystal formation; if you wait longer, the slide becomes crowded, and since the crystals are cubes, it’s hard to get them all in focus (too much relief!). I love this shot. It reminds me of Flatland.

More salt, the next day, with larger crystals.

Baking soda, which apparently creates sparry crystals.

Sugar! This one took a full day to form interesting crystals. But wow, they are gorgeous!

More sugar, almost a butterfly-like configuration. I expect the symmetry is coincidence.

The biggest surprise for me — my saliva, 24 hours later, had created these awesome fractal patterns. At first I thought it might be nucleation following tiny scratches in the slide glass, but the fact that they’re fractal renders this unlikely. No clue what this is, but my best guess is that it’s just random diffusion patterns, like these manganese fractal patterns.
Aha, I found an article titled “Dendritic growth in viscous solutions containing organic molecules” which has these great examples:

Caption: “Crystal patterns of some body fluids: (a) saliva; (b) cerebrospinal fluid; (c) urine; (d) blood serum.”
My examples seem to match the “blood serum” image the best, but I assure you it was saliva. Not sure what’s going on in their sample (a)!

The more I panned across the expanse of fractal growth, the more it started looking like a map of a European city. Is it not marvelous?
5 Comments
5 of 5 people learned something from this entry.
January 18th, 2013 at 9:39 pm (Biology, Plants)
The world just expanded by a factor of ten. At least.
Of course this is true in a literal sense, given the arrival of my Celestron 44345 microscope. I can now see down to scales previously invisible to my eye, magnifying at 40x, 100x, 200x, 1600x! But even more meaningful is the figurative way in which things have expanded. I have access to a rich, teeming layer of reality that previously existed only in a hypothetical fashion. And because this is a microscope with a digital camera embedded in it, I can also store and share what I see.
I first took a look at the seven prepared slides that came with the microscope. Here are some examples of the fantastic sights I saw (click to zoom):
Pine:
 Epithelium:
Epithelium:
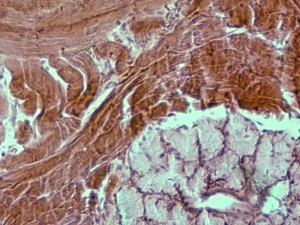 “Apple” (seed? cell? blossom? wha?):
“Apple” (seed? cell? blossom? wha?):

I’ve now placed an order for a set of blanks so that I can prepare my own slides to study anything I encounter — and even just within the confines of my house there is a veritable zoo of things to study. High on my list is sampling from the cornucopia of interesting structures that grow in my compost bin. I can’t wait to share what I discover!
3 Comments
1 of 2 people learned something from this entry.
September 8th, 2012 at 11:10 pm (Biology, Chemistry, Food)
I think I’ve finally figured out the longtime mystery about why tea both wakes me up and calms me down (anxiety-wise). I consistently feel a physical effect after drinking a cup of tea, as if a knot inside my stomach dissolves, and problems don’t seem quite as pressing, and it’s easier to be friendly and sociable. This sounds like the opposite of caffeine, which is known to increase irritability.
But just recently, I stumbled on a possible explanation. It’s not the caffeine at all! It’s something called L-theanine:
“Theanine is able to cross the blood-brain barrier and appears to have psychoactive properties. […] it appears to increase levels of the inhibitory neurotransmitter GABA, and to a lesser degree, dopamine.”
Source: livestrong.com
L-theanine is often mentioned in conjunction with green tea, but it is also found in black tea. This article says green tea has about 8 mg per cup, while therapeutic doses of the stuff (for anxiety disorders) are more like 200-600 mg. Another source indicates 15-30 mg per cup, and that “L-theanine increases the production of dopamine and serotonin, two brain chemicals associated with alertness, pleasure, and a good mood.”
Further, there’s evidence of a lack of side effects (at least in rats):
“In 2006, a study conducted on rats administered super-high doses of L-theanine daily for a 13 weeks found no consistent or significant negative effects on behavior, food intake, body weight, clinical chemistry, urine, blood, morbidity or mortality.”
Source: livestrong.com
Therefore, I should be free to experiment away. And if theanine is the active agent, then I should get the same mood benefits (though not the wake-me-up effect) from decaf green tea. If only it were easier to do controlled experiments on oneself!
1 Comments
1 of 1 people learned something from this entry.
May 4th, 2012 at 9:28 pm (Biology, Planets, Spacecraft)
The 1976 Viking landers conducted a handful of experiments that involved injecting a nutrient-laden solution into Martian soil, then measuring gases given off in response. Indeed, gases were observed from the regular soil, but not from soil that was first heated to 160 C (sterilized). That seemed intriguing to many scientists—but others noted that the same result could be obtained through (abiotic) chemical oxidation triggered by the application of water. If I understand the arguments, heating the soil would break down the presumed oxidizer in the soil so it would then react less or not at all to a new injection of moisture.
 But lo and behold, the scientists who (still) insist that Viking found life have published a new paper: “Complexity Analysis of the Viking Labeled Release Experiments” by Bianciardi, Miller, Straat, and Levin. They’ve used “complexity variables” to characterize the time series data, then clustered them (with k-means clustering, k=2). Indeed, they found that presumed “active” samples (including some examples from Earth) clustered together while presumed “inactive” samples (including some controls from Earth) clustered in a different group.
But lo and behold, the scientists who (still) insist that Viking found life have published a new paper: “Complexity Analysis of the Viking Labeled Release Experiments” by Bianciardi, Miller, Straat, and Levin. They’ve used “complexity variables” to characterize the time series data, then clustered them (with k-means clustering, k=2). Indeed, they found that presumed “active” samples (including some examples from Earth) clustered together while presumed “inactive” samples (including some controls from Earth) clustered in a different group.
Since my dissertation was on clustering, I thought I should take a look and see how this machine learning method was being used in this setting. And, well, I’m just not convinced. Yes, they do seem to have gotten two distinct populations. But they only used 15 samples (11 from Mars, 4 from Earth) and that hardly seems sufficient to characterize the range of behavior, nor are they all obviously comparable (one time series consists of “core temperature readings taken every minute from a rat in constant darkness”; how is this related to possible bacterial activity in soil? Is darkness relevant? What about a rat in daylight, or a diurnal cycle?). The authors have agreed that more data would be better. I think more data, and thoughtfully chosen, would be essential.
My other reservation is about the “complexity variables” that were used. These are presented with no justification or discussion:
- LZ complexity
- Hurst exponent
- Largest Lyapunov exponent
- Correlation dimension
- Entropy
- BDS statistic
- Correlation time
Especially since these generated the 7D space in which the clustering happened, it’d be nice to have some intuition about why these might relate to life. There are some brief comments about life being “ordered” and of “high complexity” (and I’ve worked on this subject myself!) but I’m not convinced that the distinction they found is truly meaningful.
I don’t want to be unscientifically biased or negative. The results as presented in the paper do seem to show a quantitative separation between active and inactive samples. But this should be conducted with hundreds or thousands of samples from the Earth at the very least, where we have tons of examples of life-bearing soils as well as artificial or sterilized samples. These could fill out the feature space and properly position the Viking observations in more context.
Of course, it would also be useful to get more Martian samples!
1 Comments
1 of 1 people learned something from this entry.














